Micelles are spherical aggregates made of amphililic compounds, i.e. compounds with hydrophillic and lipophillic regions. Their amphilicity means that they form hollow spheres when in aquesous solutions, such as blood or cellular fluid. By inserting drugs into the spheres center it allows the drug to be carried over lipophillic membranes such as the blood-brain barrier
A Berkley Lab team, led by Ting Xu, have developed nanocarrier micelles which they have named 3HMs, as they are colid-coil 3-helix micelles, to carry drugs for brain cancer treatments into patients brains.
Glioblastoma multiforme (GBM), which are more commonly known as octopus tumors, are one of the most aggressive forms of brain cancer. Their deadliness is a result of the tumor cells unique method of spreading into the surrounding tissue. The cancer is resistant to current treatments and is almost impossible to remove by operation.
Octopus tumours are in fact so deadly that, in most cases, the cancer becomes fatal in less than 15 months. It has been shown that each year about 15,000 people are killed by GBM in the US alone.
The blood-brain barrier is a complex network of blood vessels that acts as a filter between the body and the brain. The barrier allows nutrients to enter the brain whilst blocking other substances from reaching the highly sensitive brain tissue. Whilst the blood-brain barrier serves to protect the brain it also is major barrier for combating the disease as the barrier can prevent therapeutic drugs from accessing the brain.
As a result, a suitable technique is required to transport therapeutic drugs through this complex barrier. A team from Berkeley Lab have developed 3HM nanocarriers which could offer potential treatments for GBM.
The nanocarriers were tested on rats GBM tumors using a combination of techniques such as magnetic resonance imaging, positron emission tomography, and copper-64 (a radioactive form of copper) imagaing. It was observed that the 3HM nanocarriers can pass the blood brain barrier and collect within the GBM tumors at a higher concentration compared to any nanocarriers being currently used
Our 3HM nanocarriers show very good attributes for the treatment of brain cancers in terms of long circulation, deep tumor penetration and low accumulation in off-target organs such as the liver and spleen said Xu, who also holds a joint appointment with the UC Berkeley’s Departments of Materials Sciences and Engineering, and Chemistry.
The fact that 3HM is able to cross the blood brain barrier of GBM-bearing rats and selectively accumulate within tumor tissue, opens the possibility of treating GBM via intravenous drug administration rather than invasive measures. While there is still a lot to learn about why 3HM is able to do what it does, so far all the results have been very positive.
Ting Xu - Berkley Lab
Glial cells represent roughly 90% of the brain cells. Unlike neurons these cells experience a cell cycle of birth, separation and mitosis. Gilial cells give chemical and physical support to the neurons in the brain. However, as gilial cells are constantly dividing there is a possibility that they may become cancerous.
Once gilial cells become cancerous, they can assume different shapes, and this shape shifting means it can be difficult to detect the tumors until they become extremely large. As the glial cells have extensive tendrils the surgical removal of cancerous gilial cells is complicated. Surgery on gilial tumours has been compared to battling the legendary Hydra, with each tendril causing more cancer.
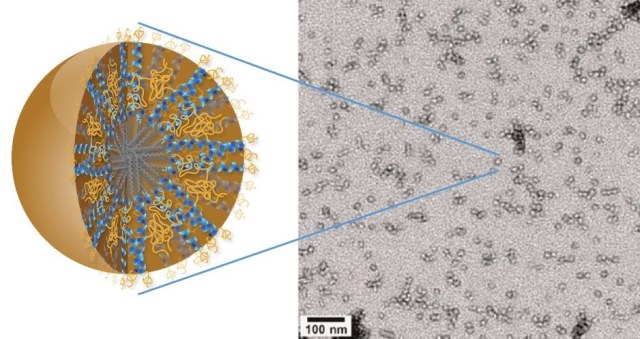 At only 20 nanometers in size and featuring a unique hierarchical structure, 3HM nanocarriers meet all the size and stability requirements for effectively delivering therapeutic drugs to brain cancer tumors. Credit: Ting Xu, Berkeley Lab
At only 20 nanometers in size and featuring a unique hierarchical structure, 3HM nanocarriers meet all the size and stability requirements for effectively delivering therapeutic drugs to brain cancer tumors. Credit: Ting Xu, Berkeley Lab
Whilst there are approved therapeutic drugs for GMB , these therapies do not improve the patient survival rate because the accumulation of therapeutic drugs in the brain is restricted by the blood brain barrier. Therapeutics drugs used for GBM treatment are usually transported across this barrier in liposomes that measure 110 nm in size, which is too large for the barriers filter. The 3HM nanocarriers measure just 20 nm in size so readily cross into the brain.
3HM is a product of basic research at the interface of materials science and biology. When I first started at Berkeley, I explored hybrid nanomaterials based on proteins, peptides and polymers as a new family of biomaterials.
During the process of understanding the hierarchical assembly of amphiphilic peptide-polymer conjugates, my group and I noticed some unusual behavior of these micelles, especially their unusual kinetic stability in the 20 nanometer size range.
We looked into critical needs for nanocarriers with these attributes and identified the treatment of GBM cancer as a potential application.
Ting Xu - Berkley La
The researchers performed systematic MRI and PET studies to learn how the size of a nanocarrier can impact the biodistribution and pharmacokinetics in rats with GBM tumors. Both liposome nanocarriers and 3HM nanocarriers were labeled with copper-64 during the studies. The study results demonstrated that the 3HM nanocarriers are effective GBM delivery vessels. They also showed that the potential nanoparticle design for brain cancer treatment can be enhanced through MRI and PET imaging of tumor kinetics and nanoparticle distribution.
The study has been reported in The Journal of Controlled Release