Jul 11 2016
A team of researchers from the University of Illinois based at Urbana-Champaign have demonstrated graphene’s doping-induced tunable wetting and adhesion feature, opening doors to unique and new possibilities for advanced coating materials and transducers.
 Doping-induced tunable wetting of graphene. (Photo credit: University of Illinois)
Doping-induced tunable wetting of graphene. (Photo credit: University of Illinois)
“Our study suggests that the doping-induced modulation of the charge carrier density in graphene influences its wettability and adhesion,” described SungWoo Nam, an assistant professor in the Department of Mechanical Science and Engineering at Illinois. “This work investigates this new doping-induced tunable wetting phenomena which is unique to graphene and potentially other 2D materials in complementary theoretical and experimental investigations.”
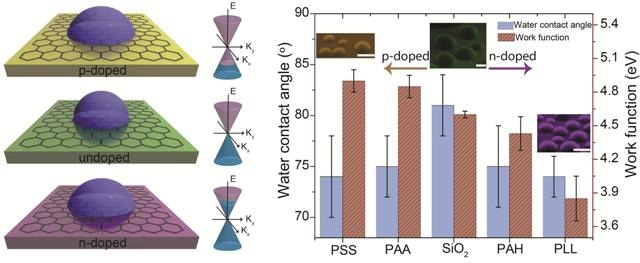
According to the researchers, graphene with its optically transparency and remarkable mechanical and electrical properties, could transform the fields of surface coatings and electrowetting displays. A material’s wettability is generally stable in the absence of external factors, and is categorized as either hydrophilic (water-loving) or hydrophobic (water-repelling), where water beads up on the surface. Based on the particular application, a choice between hydrophilic or hydrophobic material has to be made. For instance, in electrowetting displays, the hydrophilic properties of display material are improved using a stable externally impressed electric current.
The paper that appears in Nano Letters is titled,“Doping-Induced Tunable Wettability and Adhesion of Graphene.”
What makes graphene special is that, unlike conventional bulk materials, it displays tunable surface wetting characteristics due to a change in its electron density, or by doping. Our collaborative research teams have discovered that while graphene behaves typically as a hydrophobic material (due to presence of strongly held air-borne contamination on its surface), its hydrophobicity can be readily changed by changing electron density. Our study shows that graphene demonstrates tunable wettability—switchable hydrophobic and hydrophilic behavior—when its electron density is changed by subsurface charged polymers and metals (a.k.a. doping). This finding sheds lights on previous unclear links between quantum-level charge transfer and macroscopic surface wettability for graphene. This exciting finding opens new doors of possibility for tunable surface coating and electrowetting displays without continuous external electric current supply, which will translate into significant energy savings.
Ali Ashraf, Graduate Student, University of Illinois
“In addition, we investigated another closely related property—surface adhesion,” Nam said. “We observed changes in electron density of graphene leads to a change in adhesion, which determines how graphene interacts with other hydrophobic and hydrophilic molecules, which is important for graphene-based chemical and biosensors. Our finding suggests that it is possible to make reusable, self-cleaning graphene sensors that can first interact with hydrophobic molecules for detection, and then separates from them (i.e. cleans itself) by enhanced hydrophilicity via electron density modulation.”
Apart from Ashraf and Nam, co-authors include Michael Cai Wang, Yanbin Wu, Tao Sun, Keong Yong, Richard T. Haasch, Yuhang Jing, and Narayana Aluru, a professor of mechanical science and engineering, whose research team implemented theoretical modelling of this new experimental observation in this research. This study got its financial support from the Air Force Office of Scientific Research/Asian Office of Aerospace Research Development (AFOSR/AOARD), National Science Foundation CAREER Award and by Nano Bio Info Technology (NBIT) Phase III Program.
The experiments were performed partially in the Micro and Nanotechnology Laboratory, Frederick Seitz Materials Research Laboratory and the Beckman Institute Imaging Technology Group at Illinois.
The authors note that our work independently demonstrates and further supports similar findings reported in Hong et al. (DOI: 10.1021/acs.nanolett.6b01594), showing doping-induced wettability modulation of graphene.