Mar 28 2017
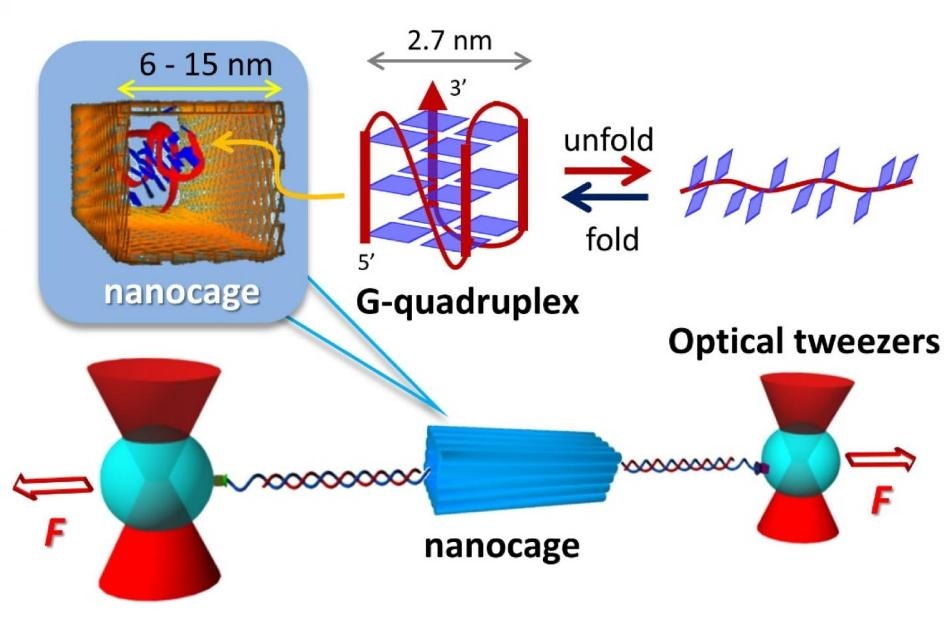 The nanocage made of DNA, surrounds a G-quadruplex, a group of guanine nucleic acids, the �G�s in the DNA sequence. The G-quadruplex molecule folds and unfolds 100 times faster inside the nanocage than without a nanocage, indicating that the nanospace stabilizes the structure. Researchers used optical tweezers to conduct the nano-scale experiment. CREDIT: Kyoto University iCeMS.
The nanocage made of DNA, surrounds a G-quadruplex, a group of guanine nucleic acids, the �G�s in the DNA sequence. The G-quadruplex molecule folds and unfolds 100 times faster inside the nanocage than without a nanocage, indicating that the nanospace stabilizes the structure. Researchers used optical tweezers to conduct the nano-scale experiment. CREDIT: Kyoto University iCeMS.
Macromolecules regularly fold and unfold themselves inside cells. The functions of these macromolecules can be determined by their various three-dimensional structures. Having an in-depth knowledge on molecule folding, complex physical processes that have an impact on allergies, cancers, and diseases can be easily understood.
G-quadruplexes are groups of various guanine nucleic acids (the “G”s in the DNA sequence) forming particular shapes resembling futuristic, three-story office buildings. For a long time, they were dismissed as not having a biological function. However, they are currently perceived to help in regulating gene expression, for diseases also.
An in-depth knowledge of the physical processes that these compounds go through while folding into extra-tight spaces might help to develop drug treatments targeting them in the future.
Carrying out experiments to study the folding process in constricted areas is highly difficult as not just the target molecule (with a length of only a few nanometers ) is easily disturbed, but the surrounding infrastructure, only a fraction larger than the target molecule, can get disturbed as well.
A research group headed by Hiroshi Sugiyama and Masayuki Endo from the Institute for Integrated Cell-Material Sciences (iCeMS) of Kyoto University worked in collaboration with researchers from Kent State University to design structures and experimental system for successfully controlling G-quadruplexes inside nanocages, also formed of DNA.
The researchers analyzed the way in which different-sized spaces have an impact on the thermodynamic stability as well as the folding and unfolding kinetics of the molecules.
“Under the confined nano-space, the G-quadruplex structures revealed unprecedented fast folding kinetics with increased mechanical as well as thermodynamic stabilities, which directly supported theoretical predictions,” concluded the research team in the research recently published in the Nature Nanotechnology journal.
The team built rectangle-shaped nanocages using DNA and wrapped the nanocages around a G-quadruplex molecule. The molecule was attached to two beads by means of tethers formed of even more DNA.
Lasers called optical tweezers were used to control the beads to exert force on the molecule, consequently causing the molecule to unfold and then refold. As the nanocage and the target both had negative charges, they repel each other similar to magnets, having no interference between them.
Small, medium, as well as large nanocages were constructed by the researchers. Compared to molecules without nanocages, the molecules in the small and medium nanocages unfolded and refolded 100 times faster.
The outcomes of the research support the predictions of other scientists in the field. However, this is the first experiment where there was no interaction between the cage and the molecule. The research team expects that the technique can also be used for analyzing other biomolecules (e.g. proteins) by making use of accurately designed nanocages.