Image Credit: Shutterstock.com/ Huen Structure Bio
The study could offer better insights on how the small protein derived from an antibody type, which occurs only in camelids (that is, alpacas, camels, and llamas) and sharks, could act against diseases such as lupus, psoriasis, rheumatoid arthritis, lymphoma and breast cancer.
This is one of a kind as researchers determine protein structure from solid samples, that is, from crystals, using X-ray or from frozen solutions with the help of electron microscopy. Yet, at times, crystals are hard to obtain, and most importantly, proteins work in the liquid state where the shape and mobility tend to change from solid-state, specifically for small species.
The NMR spectroscopy helped the researchers to map the factors relevant for nanobody function, identify any variations when the nanobody attaches to its target and prevent, as in Nb23, the formation of abnormal protein aggregation (amyloid) that causes degenerative or functional diseases.
Scientists are waiting to explore the potential of nanobodies against disease-fighting monoclonal antibodies. These are antibodies developed in the laboratory and mimic the ability of the immune system to combat harmful antigens like viruses, but they are hard to conserve and rarely penetrate solid tissues due to their size.
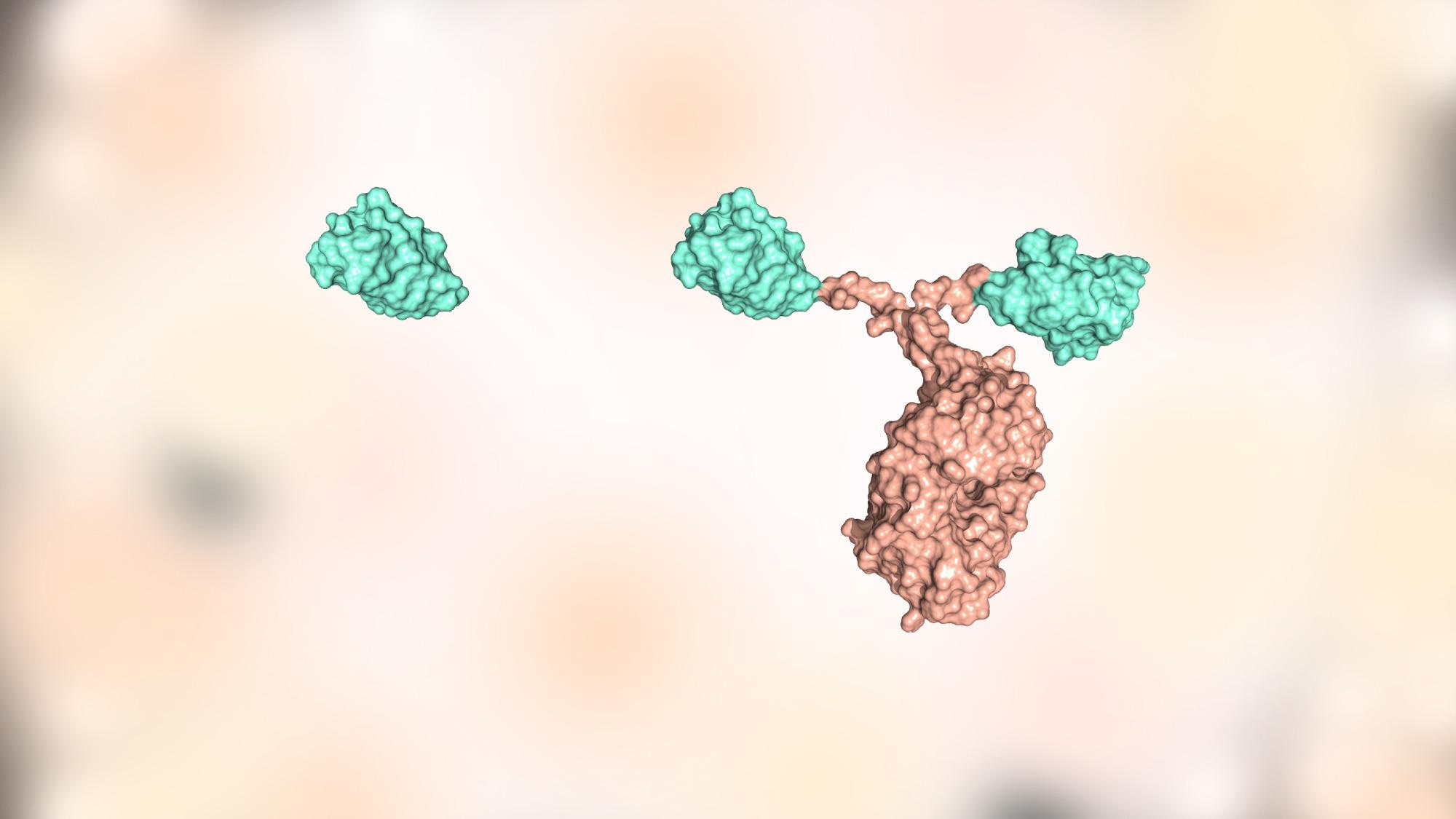
On the other hand, nanobodies are ten times smaller in size compared to antibodies and provide more stability, strong binding, good solubility and biocompatibility because of their natural origin, acting as a potential alternative for therapeutic use.
Our team is studying several nanobodies, including two in particular, Nb23 and Nb24, that bind to a key protein of the immune system called beta2-microglobulin and prevent its pathological transformation into fibrillar deposits such as those involved with degenerative or functional diseases, including Alzheimer’s, Parkinson’s and type-2 diabetes.
Gennaro Esposito, Visiting Professor of Chemistry, NYUAD
“Elucidating the structure of Nb23 and other important nanobodies is a critical step in improving our understanding of how they can bind to target proteins and help prevent the onset of these diseases,” concluded Esposito.
Journal Reference:
Percipalle, M., et al. (2021) Structure of Nanobody Nb23. Molecules. doi.org/10.3390/molecules26123567.