Zinc Tungstate – An Effective Photocatalyst
This latest research focuses on trying to recycle catalysts for multipurpose functions to minimize the expense of industrial substances for a wide variety of technical applications. The major emphasis of the work in this paper was on examining the effectiveness of the catalysis of recycled inexpensive materials for diverse purposes.
It is worth noting that substances of group VI-B (particularly tungsten) have very high efficiency in a variety of processes such as photocatalytic, electrocatalytic, and energy storage activities. Polluted water restoration is an essential approach to managing health hazards by eliminating natural contaminants from water using photocatalysis, which is among the most promising approaches for environmental cleanup.
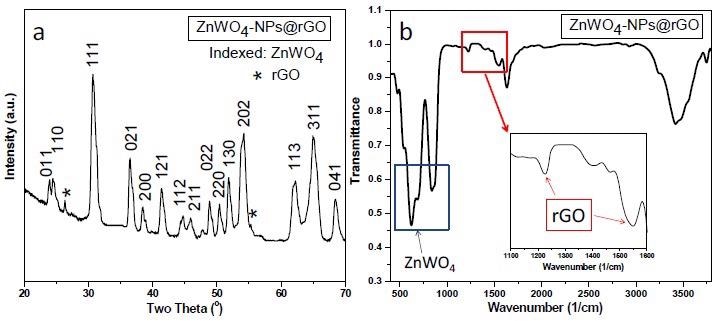
(a) XRD patterns and (b) FTIR spectrum of synthesized ZnWO4-NPs@rGO nanocomposites.© Alhokbany, N., Alshehri, S. M., & Ahmed, J. (2021).
Nanocomposites based on semiconducting materials have been studied as inexpensive, well-performing photocatalytic materials for water contamination management. Zinc tungstate (ZnWO4) possesses a high bandgap energy and is an effective photocatalyst in the breakdown of aromatic dyes under UV light.
Dyes have already been found to be decomposed by zinc tungstate nanocomposites utilized for photocatalysis. The chemical recombination of electric charges in such substances, meanwhile, poses a difficulty for research. Introducing heteroatomic structures, carbon-based compounds, or ions of transition metal into a zinc tungstate matrix may thus be beneficial for producing effective charge isolation in semiconductors and so improving the effectiveness of photocatalysis. The combination of WO3 and ZnWO4 nanocomposites has facilitated excellent opposing charge segregation, leading to their use as effective photocatalytic materials.
Nanocomposites of Zinc Tungstate and Silver
Under ultraviolet light, Ag/ZnWO4 nanocomposites have demonstrated remarkable efficiency in the breakdown of methyl orange (MO) and methylene blue (MB) dyes. Zinc tungstate nanomaterials have also been employed for a variety of functions, such as extracting heavy metals and dyes, bacterial disinfection, and photoluminescence, in addition to their usage in elements of Li-ion cells and supercapacitors. Nonetheless, some studies report that dyes are not suitable agents for visible-light breakdown processes as they absorb photons and thus suppress the abilities of photocatalytic materials. For this reason, several transition metal nanomaterials such as tungstate, gallate, molybdate, and ferrite nanomaterials have been developed for an array of uses such as treating sewage water, supercapacitor applications, and electro-catalysis.
Modifying with Reduced Graphene Oxide
Researchers are interested in surface modification of semiconductive NPs with materials having high electric conductivity and large specific areas to effectively increase the photocatalytic performance and stability of inexpensive photocatalysts. Zinc oxide nanocomposites based on graphene have already been described as potential photocatalytic materials in chemical dye breakdown processes. Hence, the team created nanocomposites incorporating reduced graphene oxide layer supplemented zinc tungstate nanoparticles (ZnWO4-NPs@rGO) for enhanced photocatalysis performance in neutral conditions. This research focused on the fabrication, analysis, and increased visible-light photocatalytic performance of ZnWO4-NPs@rGO nanocomposites.
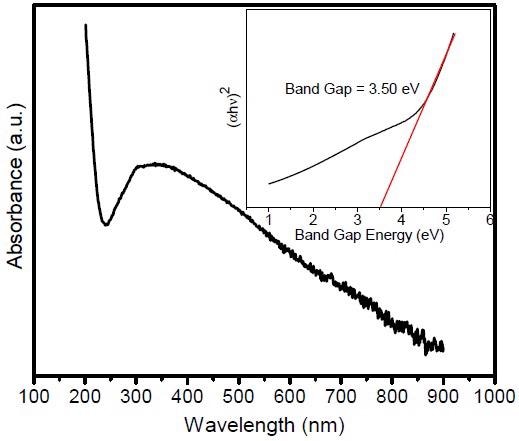
UV–Vis absorption spectrum of ZnWO4-NPs@rGO nanocomposites. Inset shows the band gap energy of ZnWO4-NPs@rGO nanocomposites.© Alhokbany, N., Alshehri, S. M., & Ahmed, J. (2021).
Can ZnWO4-NPs@rGO Photocatalysts be Recycled?
A major problem for commercial processes is the reusability of ZnWO4-NPs@rGO photocatalysts. Illuminated under visible light, the reused photocatalysis performance of ZnWO4-NPs@rGO nanocomposites was investigated for eight successive cycles. The breakdown of HMBD solutions via photocatalysis by ZnWO4-NPs@rGO nanocomposites demonstrated outstanding reused efficiency.
The researchers discovered that recycled ZnWO4-NPs@rGO photocatalysts effectively decomposed the HMBD solution for as many as eight repetitions. It should be noted that the ZnWO4-NPs@rGO photocatalytic materials were rinsed with demineralized water numerous times after every cycle before being reused for the next one. According to the findings, ZnWO4-NPs@rGO nanoscale composites perform well in photocatalysis for water treatment functions in environmental cleanup.
To Conclude
Hydrothermally produced ZnWO4-NPs@rGO nanostructured composites were found to outperform pristine zinc tungstate NPs in photocatalysis. In two hours, the photocatalytic activity of ZnWO4-NPs@rGO nanostructured composites for the breakdown of HMBD demonstrated exceptional results with about 98 percent dye breakdown, significantly superior to a lower 53 percent offered by pristine zinc tungstate photocatalysts. As a result, ZnWO4-NPs@rGO nanostructured composites have the potential to be important photocatalysts for environmental cleanup and energy purposes.
Continue reading: How Microbes are Making Nanotechnology More Eco-Friendly.
Reference
Alhokbany, N., Alshehri, S. M., & Ahmed, J. (2021). Synthesis, Characterization and Enhanced Visible Light Photocatalytic Performance of ZnWO4-NPs@rGO Nanocomposites. Catalysts, 11(12). Available at: https://doi.org/10.3390/catal11121536
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.