Because of their atomic thickness and low cross-membrane transport resistance, graphene nanoporous membranes are exciting materials for gas separation applications. The atomic thickness of graphene gives it considerable promise for separation processes.

Figure 1. Graphdiyne-based membranes. a Scanning electron microscope image of one of our membranes. Top panel shows cross-sections of the graphdiyne membrane, tilted by ∼54° to show both the quasi-2D layer (also indicated by the yellow lines and arrows) and the vertical wall/ merged microwell structures on top. Bottom panel shows the top view of the membrane. b TEM image of the membrane. The thinnest regions at the bottom of microwells appear dark and are indicated by the arrows. c TEM image of a flat region near the bottom of a microwell (low panel). Top left: Schematic of monolayer graphdiyne’s structure. Top right: Selected area electron diffraction pattern from the same region.
Production Methods and Advantages of Nanoporous Membranes
Top-down manufacturing is often used to induce nanoscale flaws in originally impervious 2D materials to form nanopores in 2D nanocrystals. Bottom-up production of thin nanoporous membranes, such as laminates formed of layered materials and multilayer films of inherently porous crystallites, is an alternate technique that may be more feasible in terms of applicability.
However, the processes driving gas penetration and segregation by these quasi-2D nanoporous membranes are poorly understood since they deviate dramatically from basic active transport models.
The cross-membrane transport barrier of typical 3D polymeric membranes is proportional to membrane thickness, restricting gas flow rates. Because of the availability of activation barriers, nanoporous membranes of graphene-based materials exhibit extraordinarily low molecular transport resistances.
Unfortunately, the existence of activation barriers also means that flow rates are suppressed exponentially. This tradeoff between permeation rates and selectivity is widely understood, and it drives the hunt for innovative nanoporous materials with optimum tradeoff properties.
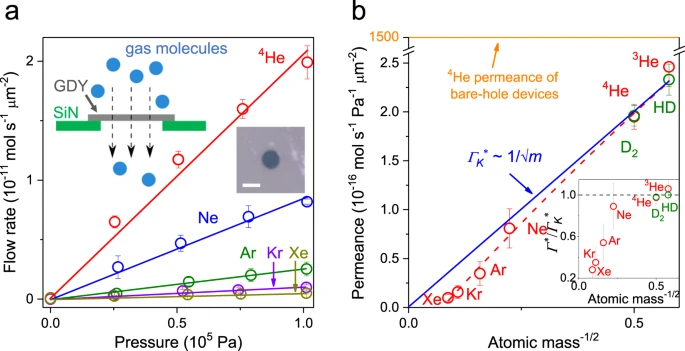
Figure 2. Gas permeation through graphdiyne-based membranes. a Examples of the measured flow of noble gases through micrometer-sized membranes (symbols). Solid lines: Best linear fits to the data. Error bars: standard deviation. Left inset: Schematic of our experimental setup. Right inset: Optical micrograph of one of our graphdiyne devices used in the experiments. The aperture is made in a 500 nm-thick silicon nitride (SiN) membrane and appears as a dark circle. It is covered by a suspended graphdiyne film (GDY). Scale bar, 2 μm. b Observed gas permeance at room temperature. Symbols are the experimental data with the error bars indicating Standard deviation using at least three different devices for each gas. The blue line shows the best fit by the Knudsen dependence using the data for light gases from 3He to Ne. Red curve: Guide to the eye. Inset shows the ratio of gas permeance to that from Knudsen dependence. For free molecular flows, the ratio is expected to be equal to one as indicated by the black dashed line. Source data are provided as a Source Data file.
Development of Novel Graphdiyne-Based Nanoporous Membranes
Graphdiyne, a carbon allotrope with inherent triangle holes a few angstroms in size, is one of the potential crystals for highly efficient molecular screening membranes. Its prospective utilization in gas separation methods has been extensively examined in theory and computations, but experimental evaluation of its gas separation qualities is still missing.
In this study, researchers explored gas transport across graphdiyne-based nanoporous membranes. The nanoporous membranes were hung above micrometer-sized pores engraved on silicon wafers to accomplish this.
The resulting membranes were sandwiched between two vacuum-tight compartments, one loaded with the gas under research and the other with a high vacuum, and were linked to a mass spectrometer.
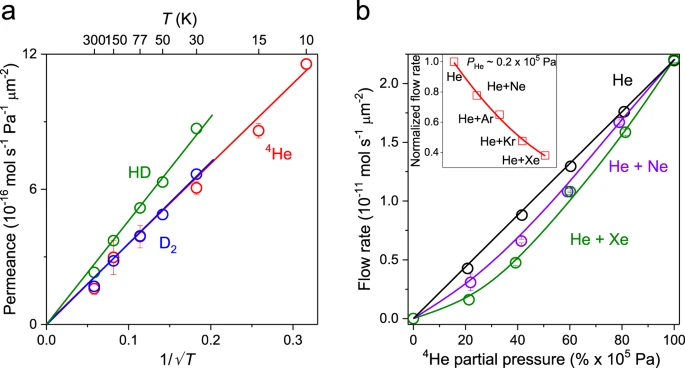
Figure 3. Knudsen and non-Knudsen gas transport through nanoscale quasi-2D pores. a Temperature dependence of gas permeance for light gases. Symbols: experimental data with the error bars indicating SD. Solid lines: best fits showing the Knudsen dependence. b 4He permeation as a function of its partial pressure within binary gas mixtures. The total pressure of the mixed gas is kept at 1 × 105 Pa. Solid curves: guides to the eye. Top inset: Helium flow rate at the partial pressure of 0.2 × 105 Pa with 0.8 × 105 Pa added by the other noble gases. The red solid line is a guide to the eye. Source data are provided as a Source Data file.
Characterization Methods for Nanoporous Membranes
Scanning electron microscopy (SEM) was utilized to examine the interconnecting nanometer-thick vertical slats and microwell topologies of the nanoporous membranes. Cross-sectional images of graphdiyne membranes were captured using a scanning electron microscopy/focused ion-beam (SEM/FIB) system.
As a consequence of re-deposition during the ion milling process, the structure of graphdiyne films along the trench looked brighter than its initial appearance. The composition of the produced nanoporous membranes was studied using transmission electron microscopy (TEM) images.
The researchers employed a handmade continuous flow cooling system to measure gas permeation at cryogenic temperatures. A transfer tube linked the cooling compartment to a liquid helium tank. When the cooling chamber is pumped out, the liquid cryogen swells via the control valve into cold gas, which flows through the tube's middle layer towards the sampling chamber area.
An outermost layer of less cold gas travels in the opposite direction from the sampling site to the exhaust and functions as a radiation barrier around the central layer. The flow velocity of cryogen (and hence the cooling rate) may be regulated by altering the vacuum pumping velocity through another valve located between the pump and the transfer tubing.
Important Findings of the Research
In contrast to the previously published atomically thin films with nanopores generated by top-down synthesis, the developed quasi-2D nanoporous membranes display comparable selectivities paired with high flow rates due to a comparatively high pore concentration.
The data also shows that the nanoporous membranes produced in this work outperform the known tradeoff constraints in terms of permeance-selectivity effectiveness. Adsorption, unlike in 2D membranes made with perforated graphene, plays a fundamentally different function in these quasi-2D nanoporous membranes.
Molecules adsorbing on graphene may readily migrate in-plane, significantly increasing penetration. However, the adsorption of complex compounds on the internal surfaces of graphdiyne-based membranes inhibits permeability.
Based on these observations, it is reasonable to conclude that if the mechanical strength of carbon allotropes with bigger unit cells, such as graphdiyne, can be obtained, they will have substantial potential for use in large-scale industrial applications requiring gas separation.
Reference
Zhou, Z. et al. (2022). Gas permeation through graphdiyne-based nanoporous membranes. Nature Communications. Available at: https://www.nature.com/articles/s41467-022-31779-2
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.