Jul 27 2009
Haoshen Zhou the Energy Interface Technology Group, the Energy Technology Research Institute of the National Institute of Advanced Industrial Science and Technology (AIST) and Yonggang Wang have developed a new-type lithium-air cell with large-capacity based on new structure.
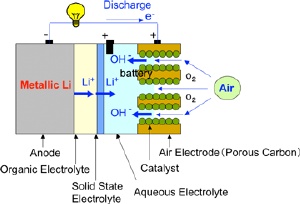 Configuration of a newly developed lithium-air cell
Configuration of a newly developed lithium-air cell
Lithium-ion batteries are widely used in mobile phones, computers and other electronic devices, however, their energy density is still insufficient for them to be used in electric vehicles. Lithium-air batteries are attractive because they are theoretically advantageous in terms of their potential for future large-capacity batteries. However, one of serious problems with lithium-air batteries reported to date is that a solid reaction product (Li2O or Li2O2), which is not soluble in organic electrolyte, clogs on the air electrode (=cathode) in the discharge process. If the air electrode is fully clogged, O2 from atmosphere cannot be reduced any more.
In this study, an organic electrolyte is used on the anode (metallic lithium) side and an aqueous electrolyte is used on the cathode (air) side. The two electrolytes are separated by a solid state electrolyte (lithium super-ion conductor glass film=LISICON) so that the two electrolytic solutions do not intermix. Only lithium ions pass through the solid electrolyte, and the battery reactions proceed smoothly. At the cathode, the reaction product in the discharge process is water-soluble and no solid substances are produced. Continuous discharging of 50,000 mAh/g (per unit mass of the carbon, catalyst and binder) has been experimentally confirmed.
This technology holds great potential for automobile batteries. At a filling station, the driver of a vehicle thus equipped could exchange the aqueous electrolyte for the air electrode and refill the metallic lithium for the anode in the form of cassettes, and then continue driving without waiting for batteries to be recharged. It is easy to retrieve metallic lithium from the aqueous electrolyte, so that lithium can be reused. This is truly a new type of lithium fuel cell.
The result of the study will be presented at an Electrochemical Society of Japan meeting held in Kyoto on March 31, 2009.