Breast cancer is the most common cancer type in women, with reports from the World Health Organization (WHO) stating the diagnosis rates in 2020 consisted of 2.3 million while the number of deaths was 685,000 globally.
Cancer is one of the most prevalent pandemics faced by the global population. While conventional therapies such as chemotherapy, radiotherapy and surgery are useful and effective, their limitations have led to research into innovative treatment strategies.
Chemotherapy drugs that consist of toxicities to eliminate cancer cells can be inefficient due to their inability to select the targeted cells as opposed to healthy cells. This can lead to a wide range of adverse effects such as hair loss, infertility as well as organ failure; additionally, surgery and radiation can cause effects such as wound infections and pulmonary complications.
The recurrence of cancer after treatment is another reason behind the requirement of innovative cancer therapeutic developments. Cancer cells become resistant to chemotherapy drugs, leading to recurrence and poor prognoses.
These limitations for conventional cancer therapies have increased the interest in the area of gene therapy as an innovative treatment option which may be the answer that solves the current challenges faced.
The efficiency of gene therapy is dependent on the vector chosen for delivery, with viral vectors being the most commonly used; however, this approach may not be the most effective due to associated issues such as safety, immunogenicity and production. This has led to alternative strategies such as non-viral modalities, which may be a safer and more effective approach for gene therapy to treat cancer.
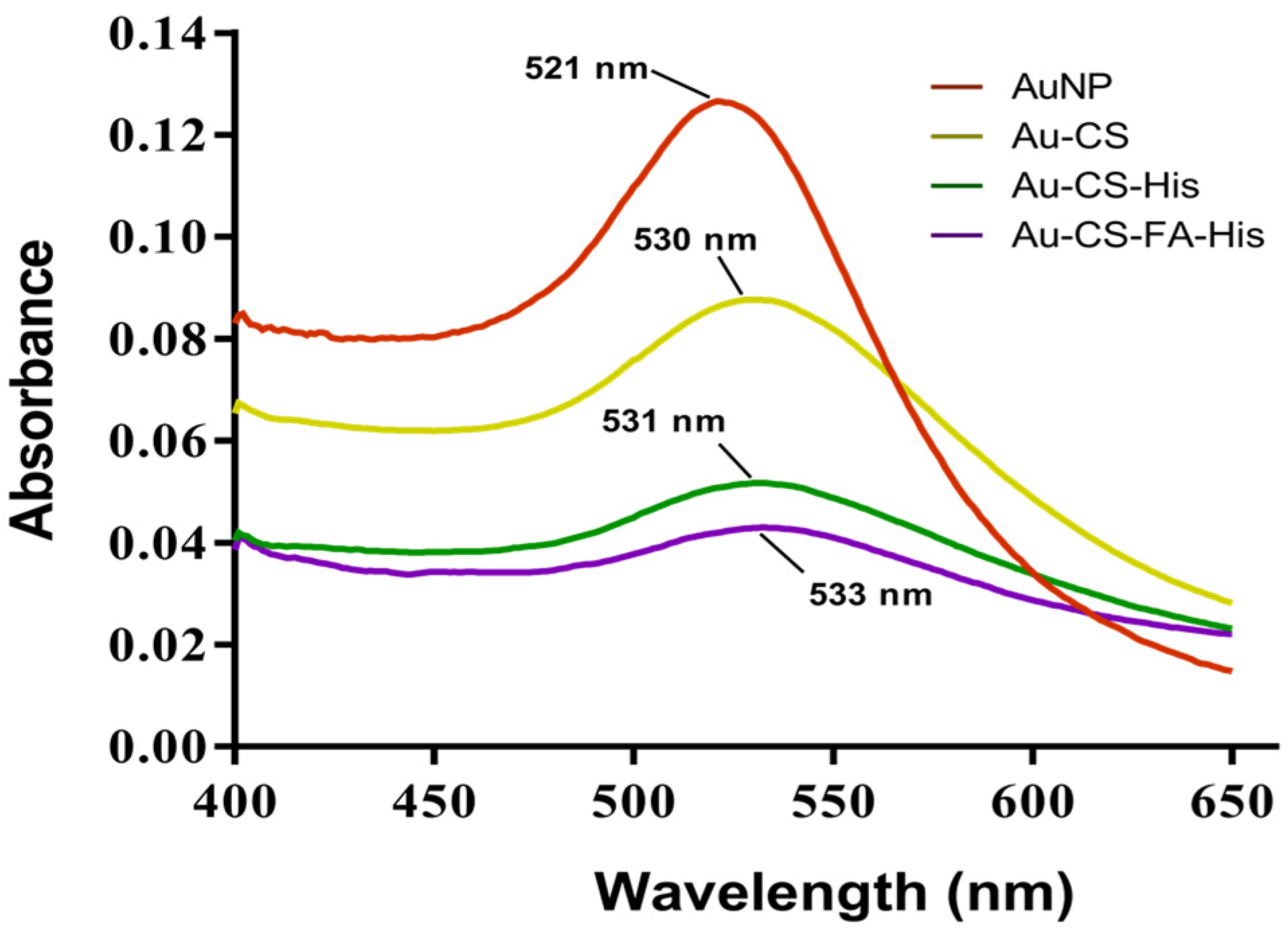
UV–Vis spectrum of AuNPs and FAuNPs. © Joseph, C., Daniels, A., Singh, S. and Singh, M., (2021)
Innovative Gold Nanoparticles for Gene Therapy
Gold nanoparticles (AuNPs) have become a significant and common incorporation of nanotechnology into medicine, and its involvement with gene therapy can only further aid in advancing the cause.
These innovative particles have remarkable physiochemical properties, with benefits such as being easy to manufacture and functionalize, easily tailorable in size, and low toxicity. The functionalization of AuNPs increases their benefits for use within gene therapy as it increases their targeting ability which will be useful for cancer treatment.
Functionalization of gold nanoparticles with chitosan (CS), a non-toxic, natural, biodegradable polysaccharide enables the coating of nanoparticles due to the interaction between the active amino groups and negatively charged metal nanoparticles. Additionally, the incorporation of histidine (His), a polar amino acid with a unique structure is recognized as being one of the strongest binders to metal ions including gold due to its carboxylic, amino and imidazole groups.
Existing research investigating His interaction illustrates the potentiality of increasing efficiency of therapeutic delivery with this approach due to the imidazole ring that can disrupt and destabilize cells and endosomal membranes. The inclusion of histidine on gold nanoparticles could potentially aid in delivering therapeutic agents.
Targeting nanoparticles to specific areas such as through specific targeting ligands can be used to target the folate receptor (FR) which has become a useful target for tumour specific delivery of genes and drugs due to their overexpression, a key difference which separates nonproliferating healthy cells which lack FR expression.
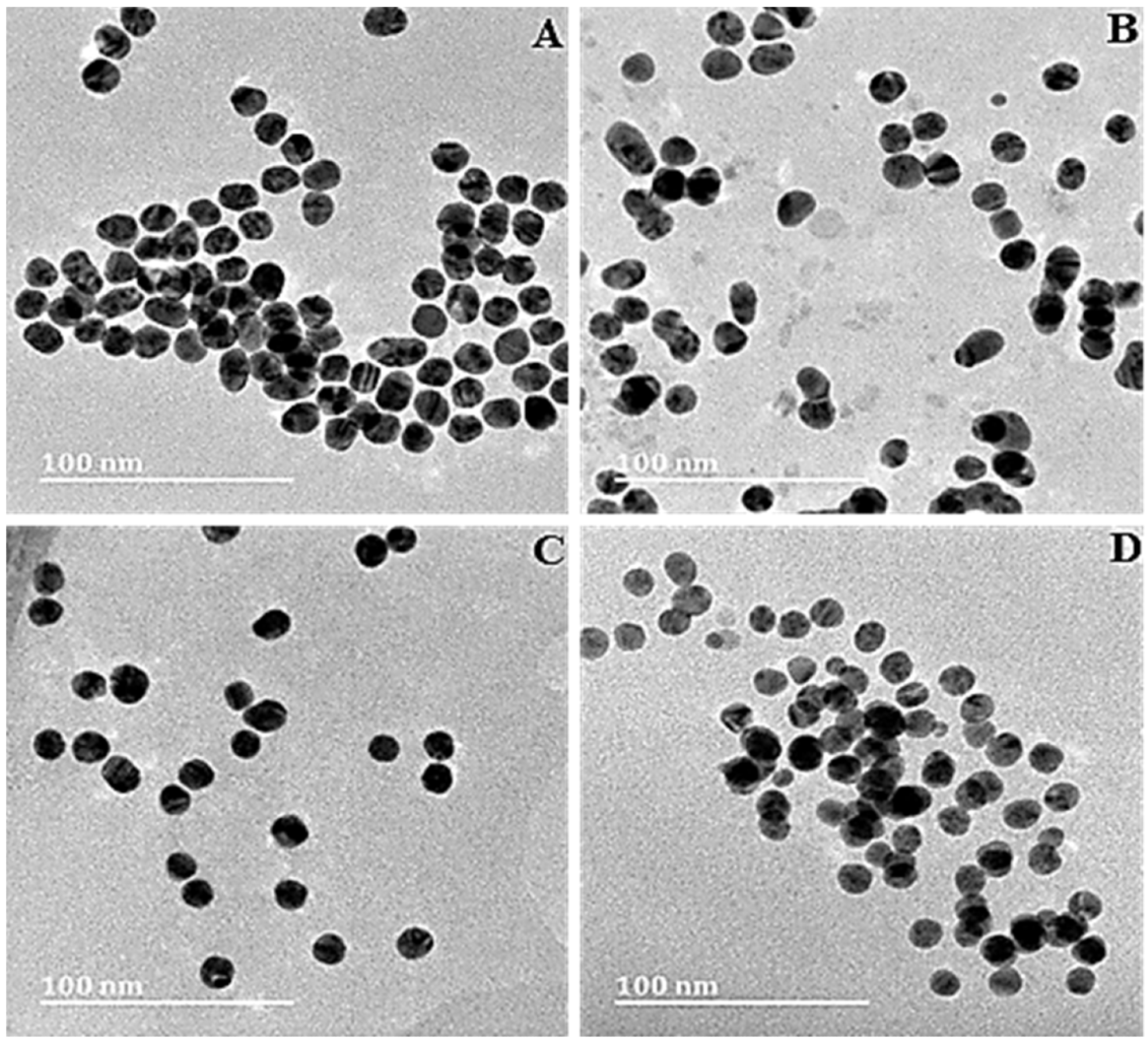
HRTEM images of (A) AuNPs, (B) Au-CS, (C) Au-CS-His and (D) Au-CS-FA-His. Scale bar = 100 nm. © Joseph, C., Daniels, A., Singh, S. and Singh, M., (2021)
This innovative research involving the fabrication of an efficient gene delivery vehicle utilizes gold nanoparticles, functionalized with chitosan, histidine and folate in order to deliver pCMV-Luc-DNA (pDNA) reporter gene to breast cancer cells. Intriguingly, this research aimed to create a perfect system to target breast cancer cells, starting as a proof-of-concept experiment with in vitro studies, with functionalization which provided more stability and specificity for targeted treatment.
The research team found this approach protected pDNA cargo from nuclease digestion. This is significant as effective transport would lead to enhanced transgene expression, and so the safe transport of the pDNA is critical before release upon arrival of the targeted area. However, with further studies illustrating pDNA still being bound to the functionalized gold nanoparticles and being retained in the wells, such strong interaction may require further optimization as it would mean the pDNA could not be effectively released from the nanocomplex due to the lack of efficient break down.
The team looked at different breast cancer cell lines such as including a human embryonic kidney cell line and two human breast cancer cell lines, to test cytotoxicity, a very significant component of therapeutic delivery, to ensure safety of this procedure for in vivo studies. They found this approach to be a fairly safe method of transporting the reporter gene, a promising step for targeted therapies.
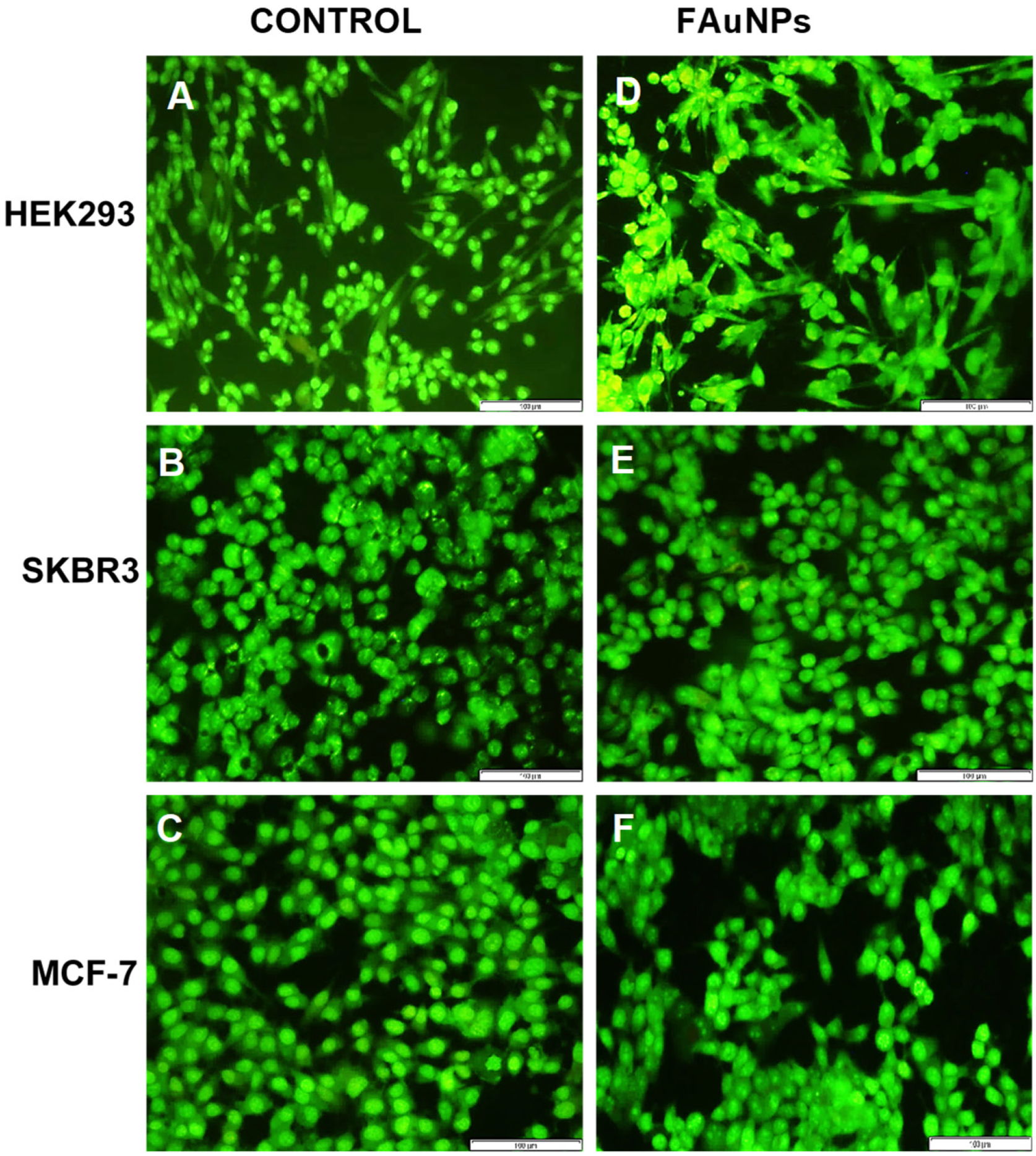
Fluorescent images from the acridine orange/ethidium bromide apoptosis assay in the HEK293, SKBR3 and MCF-7 cell lines at 20× magnification. Control = untreated (A) HEK293 (B) SKBR3 and (C) MCF-7 cells. FAuNP treated cells (D) HEK293 cells with Au-CS at the suboptimum ratio, (E) SKBR3 cells with Au-CS-FA-His at the supraoptimum ratio and (F) MCF-7 cells with Au-CS at the optimum ratio. Scale bar = 100 µm. © Joseph, C., Daniels, A., Singh, S. and Singh, M., (2021)
Future Outlook
Gold nanoparticles have significant applications in medicine and are a strong contender for use as a non-viral delivery agent. With their ability to be functionalized, they can be seen as the most versatile delivery complex.
This research group aimed to solve two common challenges in gene therapy, including targeted gene therapy as well as endosomal escape. The solutions for both of these lay within the functionalization of gold nanoparticles, which can be functionalized with ligands to target folate receptors overexpressed in cancer cells as well as stability provided by chitosan and histidine.
Transgene expression was found to be higher with safer delivery of the reporter gene and this can only be a step forward for targeted gene therapy for breast cancer. While this proof-of-concept study may have limitations, it provides the basis for further research which could be utilized for furthering the field of cancer therapy for breast cancer, the most common type of cancer found in women.
Continue reading: Semiconducting Polymer Nanomaterials for Cancer and Tumor Treatment.
Reference
Joseph, C., Daniels, A., Singh, S. and Singh, M., (2021) Histidine-Tagged Folate-Targeted Gold Nanoparticles for Enhanced Transgene Expression in Breast Cancer Cells In Vitro. Pharmaceutics, 14(1), p.53. Available at: https://doi.org/10.3390/pharmaceutics14010053
Further Reading
Who.int. (2022) Breast cancer. [online] Available at: https://www.who.int/news-room/fact-sheets/detail/breast-cancer
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.