Mar 4 2008
For centuries, engineers have bent and torn metals to test their strength and ductility. Now, materials scientists at the University of Pennsylvania School of Engineering and Applied Science are studying the same metals but at nanoscale sizes in the form of wires a thousand times thinner than a human hair. This work has enable Penn engineers to construct a theoretical model to predict the strength of metals at the nanoscale. Using this model, they have found that, while metals tend to be stronger at nanoscale volumes, their strengths saturate at around 10-50 nanometers diameter, at which point they also become more sensitive to temperature and strain rate. Such prediction of different strength regimes of nano-solids is important for future application and engineering design of nanotechnology.
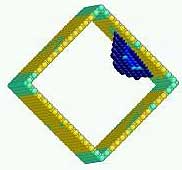 Atomic configuration of nucleation (blue atom group) in the surface layer of a square copper nanowire (yellow and green atoms) under uniaxial stress. Nucleation occurs at a partial dislocation in the surface layer. Colors refer to the breaking of local inversion symmetry.
Atomic configuration of nucleation (blue atom group) in the surface layer of a square copper nanowire (yellow and green atoms) under uniaxial stress. Nucleation occurs at a partial dislocation in the surface layer. Colors refer to the breaking of local inversion symmetry.
Such small-volume materials with relatively large surface areas are now routinely employed in microchips and nanoscience and technology, and their mechanical properties can differ vastly from their macroscale counterparts. Typically, smaller is stronger. A gold wire 200 nanometers in diameter can be 50 times stronger per area than centimeter-sized single-crystal gold. Engineers investigated the "smaller is stronger" trend.
Ju Li, an associate professor in the Department of Materials Science and Engineering at Penn, and his collaborators at the Georgia Institute of Technology have combined transition state theory, explicit atomistic energy landscape calculation and computer simulation to establish a theoretical framework to predict the strengths of small-volume materials. Unlike previous models, their prediction can be directly compared with experiments performed at realistic temperature and loading rates. This research appeared as a cover article in Volume 100 of Physical Review Letters.
Their study demonstrated that the free, exterior surface of nanosized materials can be fertile breeding grounds of dislocations at high stresses. Dislocations are string-like defects whose movements give rise to plastic flow, or shape change, of solids. In large-volume materials, it is easy for dislocations to multiply and entangle and to maintain a decent population inside; however, in small-volume materials, dislocations could show up and then exit the sample, one at a time. To initiate and sustain plastic flow in this case, dislocations need to be frequently nucleated fresh from the surface.
Since surface is itself a defect, researchers asked to what degree the measured strength of a small-volume material reflects surface properties and surface-mediated processes, particularly when the sample size is in the range of tens of nanometers. Li and his team modeled tiny bits of gold and copper to investigate the probabilistic nature of surface dislocation nucleation. The study showed that the activation volume associated with surface dislocation nucleation is characteristically in the range of 1–10 times the atomic volume, much smaller than that of many conventional dislocation processes. Small activation volumes will lead to sensitive temperature and strain-rate dependence of the critical stress, providing an upper bound to the size-strength relation.
From this, the team predicted that the "smaller is stronger" trend will saturate at wire diameters 10-50 nanometers for most metals. For comparison, computers now contain microchips with 45 nanometer strained silicon features. Associated with this saturation in strength is a transition in the rate-controlling mechanism, from collective dislocation dynamics to single dislocation nucleation.