This research can hold a significant role in advancing antimicrobial treatment, especially in the height of microbial resistance.
Bacterial Nanocellulose
Bacterial nanocellulose, which consists of cellulose, one of the most widely found compounds, is a natural biopolymer that is produced by microorganisms, such as Komagataeibacter xylinus.

NBC cultured in glass vessel took form of cylinder of 50 mm diameter. The upper panel shows the top, while the lower panel the side plane of 3 days old NBC carriers subjected to K. xylinus cells full removal (NaOH, alkaline lysis) or to process of auto-claving in water (H2O), or NBC which was not subjected to any cleansing process (not-treated, NC). © Zielińska, S., et al., (2021)
After the removal of K. xylinus, enabling BNC to be cell-free, this biopolymer can be highly useful in a range of applications without cytotoxic or general adverse effects from live tissue interaction. Its environmentally friendly properties are a key contributor to its popularity among researchers in various fields, from biomedical to biotechnology.
Interestingly, cellulose is one of the most commonly found compounds on Earth. With enhanced properties that can compete with plant cellulose, BNC retains benefits such as having a higher degree of purity, polymerization as well as water-related properties. This very useful biopolymer has already been established within literature through use within wound dressings, food additives, drug carriers, and even environment-friendly clothing.
While its establishment has spanned different areas, innovative researchers have studied its novel use as a plant cell carrier to produce pharmacologically active metabolites.
The Need for Novel Antimicrobials
With the ever-increasing concern of antimicrobial resistance for antibiotics and antiseptics, as well as the transference of resistance genes, researchers have undertaken advanced strategies to combat this crisis.

The C. majus cellular aggregates (oval shapes) on the 3-day-old NC fibrinous mesh. (A): magnification 10,000×; (B): 30,000×; (C): 50,000×, (D): close-up on C. majus cells, 100,000×. Microscope SEM Zeiss Auriga 60. © Zielińska, S., et al., (2021)
This clinical demand for novel antimicrobials as well as administration routes has led to a proof-of-concept experiment pioneered by research published in Materials. The innovative researchers aimed to investigate the possibility of culturing C. majus cells on a BNC carrier.
C. majus or celandine, is a medicinal plant that contains a large number of alkaloids such as chelidonine, chelerythrine or Sanguinarine. Traditionally, this plant was used to improve eyesight and, more recently as a method to provide mild sedation. It has also been established in applications such as treating bronchitis, asthma, and jaundice.
Its anti-inflammatory and antimicrobial effects have been investigated by researchers to combine these potentially harmful effects against microbial pathogens and have been analyzed for their efficacy within BNC carriers.
The proof-of-concept research experiment included three forms of BNC which were tested comprising:
- Unpurified polymer consisting of live K. xylinus cells
- BC with killed but not removed K. xylinus cells (partially cleaned)
- BC with killed and removed K. xylinus cells (fully purified with no bacterial cell remains)
These various BNC types were inoculated with suspended C. majus cells and then the functionalized BNC were then analyzed for the presence of C. majus metabolites such as through liquid chromatography–mass spectrometry (LC-MS). Additionally, further studies were carried out on the effect of selected BNC composites on immune system cells and microbials in vitro to comprehend the potential proinflammatory irritating activity within neutrophil cells in humans.
The results of this study found 3-day-old, unpurified BNC to have the highest content of specialized plant metabolites (C. majus) with the most complex composition of pharmacologically active substances.
Its critical antimicrobial effects were also tested against pathogens such as: Staphylococcus aureus, Pseudomonas aeruginosa and Candida albicans where the innovative biopolymer displayed antimicrobial and antibiofilm activity.
Achieving Proof-Of-Concept and Future Translation
This proof-of-concept experiment can be seen as a success with positive data involving the culture and release of pharmacologically active substances through the use of bacterial nanocellulose as an innovative carrier.
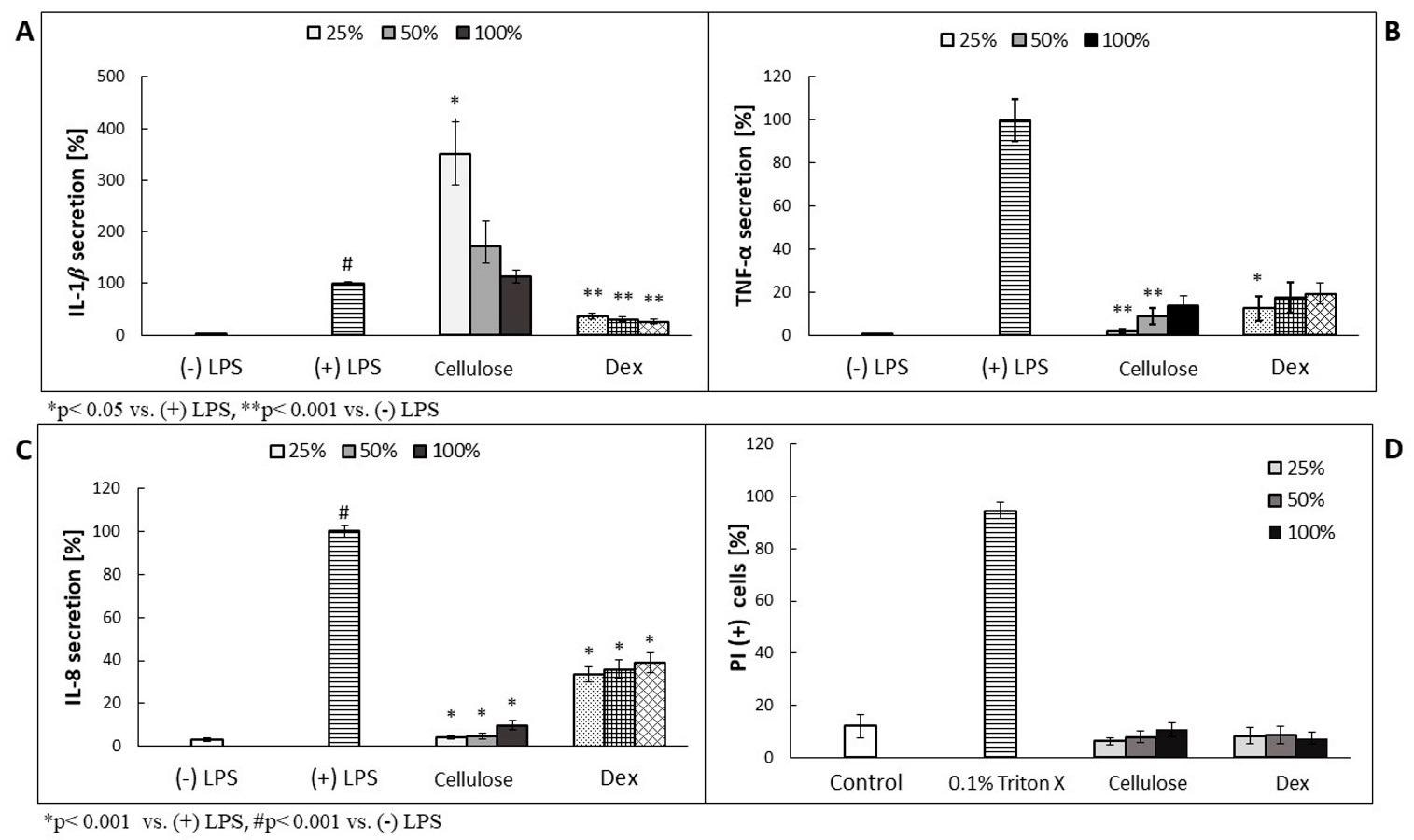
(A–C): The effect of 3-day-old NC cellulose containing C. majus cells on neutrophil IL-1β, TNF-α and IL-8 release by LPS—stimulated (100 ng/mL) cells ((+) LPS). Dex—dexamethasone at concentrations of 0.25, 0.5 and 1.0 μM; (D): The cytotoxic effect of 3-day-old NC cellulose containing C. majus cells (after four weeks of culture) on neutrophils expressed as a percentage of PI (+) cells; Control, (+) LPS; cellulose = 3-day-old NC cellulose containing C. majus cells (after four weeks of culture) The data are expressed as the mean ± SE from three donors assayed in triplicate. Statistical significance of the cellulose versus the stimulated control at p ˂ 0.001 vs. (-) LPS is marked by ** (A,B), vs. (+) LPS by * (C), and vs. (-) LPS by # (C); at p < 0.005 vs. (+) LPS is marked by * (A,B) (Mann–Whitney’s U- U-test). © Zielińska, S., et al., (2021)
While this research has just passed its proof-of-concept stage, the potentiality can be revolutionary against resistant microbial pathogens.
With further research into optimization such as the depth of infiltration of plant cells into the BNC matrix, as well as potential in vivo experimentation, this research could be a stepping-stone for novel pharmacological strategies.
This novel strategy would be welcome for combatting a global public health concern: microbial resistance against antibiotics, and its promising application can be utilized through further research into flexible composites used against microbial pathogens. This can be possibly translated into an occlusive dressing which can aid in combatting local surface infections and microbials through a plant metabolite-enriched BNC.
Continue reading: Analyzing Novel Orthodontic Composite Resins with Nanoparticles for Antibacterial Properties
Reference
Zielińska, S., et al., (2021) Bacterial Nanocellulose Fortified with Antimicrobial and Anti-Inflammatory Natural Products from Chelidonium majus Plant Cell Cultures. Materials, 15(1), p.16. Available at: https://www.mdpi.com/1996-1944/15/1/16/htm
Further Reading
Jozala, A., et al., (2016) Bacterial nanocellulose production and application: a 10-year overview. Applied Microbiology and Biotechnology, 100(5), pp.2063-2072. Available at: https://doi.org/10.1007/s00253-015-7243-4
Aronson, J. and Meyler, L., (2016) Meyler's Side effects of drugs. Elsevier, pp.468-469. Available at: https://doi.org/10.1016/B978-0-444-53717-1.01219-1
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.